 
Lewis Structure is also known as an electron-dot structure since it uses dot notations to represent the valence shell electrons in the skeletal diagram. Now, we will place the valence electrons around the atoms. We have placed the chlorine atom in the center and the fluorine atoms surrounding it. The element having the least electronegativity value is usually the central atom and thus chlorine here becomes the central atom. Among Cl and F, Cl is more electropositive than F. We will now find out which element will take the central position in the molecule. The total number of valence electrons ina ClF5 molecule = 7 + 7*5 = 7 + 35 = 42. Each one of the six atomic elements has 7 valence electrons. Both chlorine and fluorine belong to the group of halogens and therefore present in group 17. Valence electrons are the outermost shell electrons of an element that we can determine from the atomic number and Periodic table.Ī molecule of chlorine tetrafluoride has one atom of chlorine and five atoms of fluorine. Now, we will find out the suitable Lewis Structure diagram for our molecule, ClF5.Īt first, we will calculate the total number of valence electrons inside the molecule. It gives us a diagrammatic representation of the arrangement or distribution of electrons around the constituent atoms to help us have a simplified idea of bond formation. Lewis Structure is a step-by-step procedure to draw a two-dimensional sketch of a molecule or ionic structure. 
If we want to understand the science behind the chemical bonding of any given molecule, we have to first draw the Lewis Structure diagram. Let us now learn the chemical bonding inside a molecule of chlorine tetrafluoride in detail. We have several types of bonds – ionic, covalent, metallic, hydrogen, and so on. The study of atomic attraction that results in product formation is known as chemical bonding and the bond formed is called a chemical bond. ISSN 0368-1769.ClF3 + F2 -> ClF5 (at high temperature and high pressure)Ītoms of similar or different elements come together and form a new molecular composition, which creates new chemical compounds. Journal of the Chemical Society (Resumed): 3681. "The Viscosities of Iodine Pentafluoride and Ditellurium Decafluoride". Journal of the American Chemical Society. "Dielectric Constants of Liquid Chlorine Trifluoride and Iodine Pentafluoride". "Iodine Pentafluoride, Freezing and Boiling Point, Heat of Vaporization and Vapor Pressure-Temperature Relations". "The Vibrational Spectra and Structures of Iodine Pentafluoride and Heptafluoride". "Rearrangement of Amides with Iodine Pentafluoride". Journal of the Chemical Society: 2789–94. "Preparation and Crystal Structure of Osmium Pentafluoride". Zeitschrift für anorganische und allgemeine Chemie (in German). Zeitschrift für Anorganische und Allgemeine Chemie (in German). "Fluorierung von Verbindungen des Kohlenstoffs (Benzol und Tetrachlormethan mit Jod-5-fluorid, sowie Tetrachlormethan mit Fluor) ". "Crystal structure of Iodine Pentafluoride at -80°". Primary amines react with iodine pentafluoride forming nitriles after hydrolysis References For example, the reduction of osmium hexafluoride to osmium pentafluoride with iodine is conducted in a solution in iodine pentafluoride: 10 OsF 6 + I 2 → 10 OsF 5 + 2 IF 5 It has been used as a solvent for handling metal fluorides. 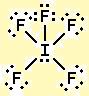
Upon treatment with fluorine, it converts to iodine heptafluoride: IF 5 + F 2 → IF 7 IF 5 reacts vigorously with water forming hydrofluoric acid and iodic acid: This exothermic reaction is still used to produce iodine pentafluoride, although the reaction conditions have been improved. It was first synthesized by Henri Moissan in 1891 by burning solid iodine in fluorine gas. It is used as a fluorination reagent and even a solvent in specialized syntheses. It is a colorless liquid, although impure samples appear yellow. Iodine pentafluoride is an interhalogen compound with chemical formula IF 5.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
